First of all, I want to remind everyone that I’m not a doctor, pharmacist, researcher, scientist, or anything of that sort, so don’t take any of my words as absolute truths.
“How to become a human guinea pig.” The idea itself seems a bit frightening and pretty risky. Who would expose their own body to substances that have never (or almost never) been tested on other humans?
Well, before judging those people as a bunch of crazy people, go to your bathroom and take a look at your medicine cabinet (if you have one…my apartment doesn’t).
Imagine that every single one of these medicines had to be tested on someone (or several someones) before being sold. All of them! 100%.

No, not this kind of guinea pig. Picture by Andy Miccone, licensed under CC0 1.0
So, we can say that if it wasn’t for those people like myself who volunteered to test drugs to see if they were safe, there wouldn’t be any approved drugs on the market (here, we’ll ignore the fact that in the past, medicines were tested on slaves, prisoners, and soldiers without their knowledge or consent).
Okay. I’m not saying that I’m the savior of the country or that I tested drugs to help humanity. It was honestly for money….a bit out of curiosity….and a bit of just messing around…although it does feel good to know that I might have helped in the development of a new drug that will eventually help some people out.
But like I said above, without people like myself who were willing to have their bodies used for research, there wouldn’t be new medicines and new treatments.
What are these medical tests where new “drugs” are tested? How are they normally done?
Truthfully, it’s a long process for a drug to become commercialized. First, it’s developed and tested in labs using computers, chemical tests, and other techniques. Afterward, it’s tested on animals to be able to predict some of the reactions that could happen when given to humans.
Passing these two stages comes the phase of testing on humans. And this is the part that interests us. This phase is divided into 4 parts:
- The researchers first test the new drug or treatment on a small group of people to check its safety, determine if the dosage is safe, and identify (adverse) side effects. Normally, 20 to 80 subjects are used. I’ve participated in this phase.
- The drug or treatment is given to a larger group of people to see if it’s effective and to better evaluate its safety. Placebos are also used. 100 to 300 subjects are tested. I’ve also participated in this phase.
- The drug or treatment is given to an even larger group of people to confirm its efficacy, monitor its side effects, compare it with commonly-used treatments, and gather information that will allow the medication or treatment to be used safely. In this case, the volunteers have a sickness that the drug can be used to combat. This way, it really evaluates the drug’s effectiveness. From 800 to 8,000 are tested. Thankfully, I’m healthy so I’ve never participated in this phase.
- Studies are done after the drug or treatment are commercialized to collect information about the effects of the drug on different populations and any side effects associated with long-term use. Thousands of people over the course of several years are tested.
So, the objective, if you are healthy, is to participate in one of the first two steps. Otherwise, if you are diagnosed with a “problem”, participate in one of the other two phases for a drug that specifically treats your problem.
My bed during one of the trials
What made me think of participating in medical trials?
To be honest, I had never thought about doing something like this before. But, as always, the idea came to me while on one of my trips.
It was during a backpacking trip that I did with a friend around Europe. We were in a hostel in Edinburgh, Scotland and I saw a guy that had one of those little hospital bracelets on his wrist. We asked him if he was sick or was in the hospital. He told us that he had just finished a medical trial and was still “under observation.”
We were intrigued right away and asked a bunch of questions like how it went, if he felt something, why he did it, if he was afraid and plenty more. Basically, he said that after testing the medicine, he had a bit of a headache, but nothing more. That he felt fine and that he, basically, tested drugs to make money.
I thought it was very interesting and the idea stuck…”maybe one day,” but without any set plans.
How did I begin testing drugs?
A few years later, I went to live in Australia. I had just arrived and was looking for a job. I researched all – seriously ALL – the possible option to make some money. I ended up finding an ad from GSK (GlaxoSmithKline) looking for healthy candidates for their database to participate in future medical trials and diverse research.
Since I had nothing to lose and I needed money, I filled out the form online, without any big expectations.
During all this online research, I also came across the idea of a mystery shopper. Something that I did, made money, got free stuff, and thought was really cool. If you want to learn more, just take a look at this article.
After a while, when I was already working, GSK called me asking if I wanted to participate in a study. They needed healthy subjects without any medical complications.
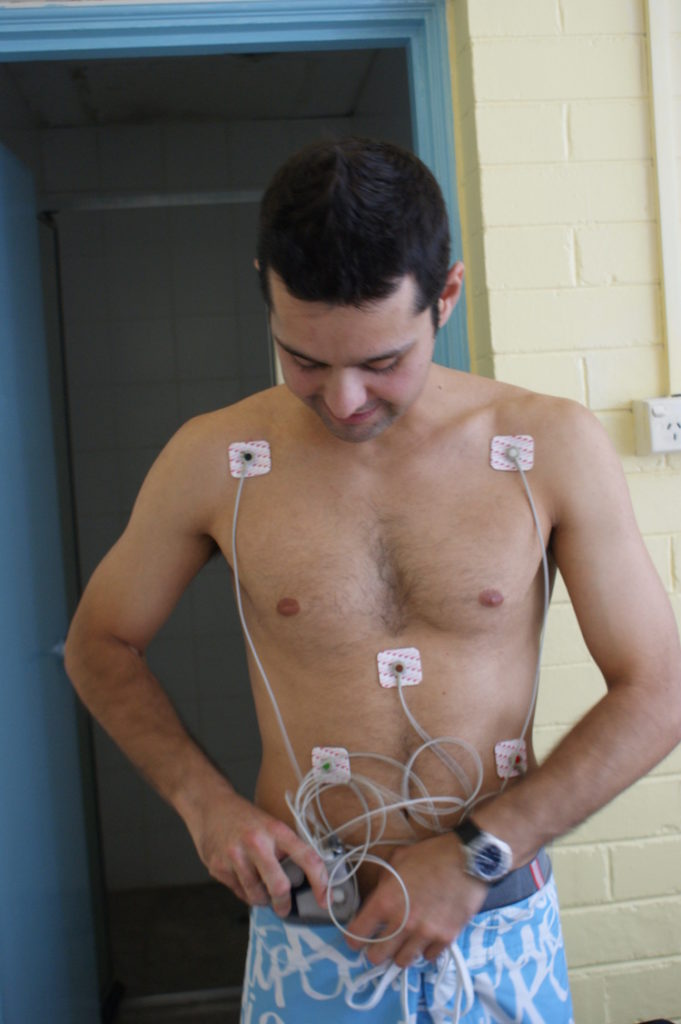
Before one of my medical trials, I had to spend 48 hours hooked up to this machine outside the hospital
What is the actual process like to participate in medical trials?
Well, each test or study for a new drug or vaccine will have different characteristics. These characteristics will be put in a summary brief and given to all potential volunteers.
This brief should have the following information:
- What type of medicine is being tested
- What type of disease it’s trying to treat
- Possible side effects
- The exact tests that will be done during the procedure
- How much blood will be taken during the process
- How many hospital visits are expected before, during, and after the test itself
- How many nights you’ll be expected to spend in the hospital or research center
- What the planned schedule is for the research
- Food, drinks, supplements, and other activities that should be avoided during the process
- Other general information about consent and legal elements of the process
Once you have applied for one of these trials and are called to do an aptitude test (to see if you are a good candidate for the trial), you’ll go to a research center or hospital and do a bunch more tests.
These tests normally include:
- A variety of blood tests
- Blood pressure
- Heart rate
- Electrocardiogram
- And a ton of others that will be specified on the information brief mentioned above
Once this is done, you’ll have to wait until they give you good or bad news about whether you can participate.
If the news is positive, you will have to go to some appointments during the day or even spend a few nights in a medical center (obviously with free food and accommodation), depending on the test itself.
The time spent in the hospital during the medical trial is pretty relaxed. Minus the time when you have to do the exams, the rest of the time is yours to read books, play video games, watch movies, or use your computer. Like a relaxing spa.
In my case, for the last medical trial I did, I remember that I spent the majority of my time planning my trip to Southeast Asia (which I paid for entirely with money from the tests) and deciding which diving school in Thailand I would do my Divemaster Trainee at. (PS: I wrote about how to become a Divemaster trainee here.)
After the entire procedure, you’ll probably have to go to the medical center a few more times to do the last check-ups to make sure everything is okay and see the evolution of the drug’s presence in your body.
After this, just wait and if all goes well, they’ll pay you for your participation.

Walking with my “girlfriend” attached to my nose during my 8-day trial in the hospital
Can anyone volunteer for medical trials?
As I mentioned above, this will depend on what phase of testing the drug is in.
If it’s in phase 1 or 2, where it hasn’t be tested on humans before, only healthy individuals can participate.
If it’s in phase 3 or 4, the focus is on individuals that have the specific illness that the medicine or vaccine wants to help with. So, it’s up to you to research your illness and see if there are any medical tests for new drugs that aim to help with it.
Pros and cons of testing medicines as a guinea pig
Pros
- You will, literally, be helping with the advance of science for humanity. Let’s be real here – if there weren’t volunteers to test new drugs or vaccines, we wouldn’t have access to many of the medicines we have today.
- Money!! You can really can make good money doing medical trials and invest in something for your future.
I met people in Australia who paid for their university with medical trials or did one trial and then left with the money earned to travel the world! - Free medical check-up. To participate in medical tests in phases 1 and 2, you have to be completely healthy. So, to see if you are qualified for this specific medical trial, you’ll have to undergo a bunch of free tests, which are normally very expensive. In case you have a health problem that you didn’t know about, you’ll be told and you can take measures to take care of yourself. I really liked these check-ups because I don’t go to the doctor much…but with these experiments, I always knew I was healthy.
- Have time to work on other projects. In many of the tests I did, I had to spend some time in the hospital. This gave me free time to work on my computer, study for my school tests, or plan a trip that I was going to take with the money I was going to make with the medical tests.
- Have access to medicines that could cost a fortune! Drugs for cancer or treatments for other diseases can cost a fortune. But by participating in a medical trial to test these specific drugs, you’ll have access to it free, which is great in case you can’t get it another way.
- You choose when you want to participate. There’s no contract saying that you do or don’t have to volunteer for a certain medical trial. Normally, research centers post available trials that need volunteers and you decide if you want to participate or not. But, if you confirm your participation and start the process and quit before the entire study finishes, no money for you!! Which brings us to the cons…

Why not a poker game to pass the time? (Notice the IVs in our arms…)
Cons
- Some trials can be very long and have many visits, and you have to participate for the whole thing. Therefore, you have to be very organized and responsible because studies can extend several months with many hospital visits. And as mentioned above, if you don’t complete the entire process, no money for you!
- You’ll have to watch your diet and exercise before, during, and after the tests. As the tests are based around your body’s reaction to certain drugs, the ingestion of certain foods, the use of certain supplements (like vitamins) and other medications, and even exercise, can affect the process. So, you will have to be willing to change your habits to adapt to the tests. Since I like to do a lot of sports, this was one part that most bothered me the most.
- You will be “stuck” in a research center for, possibly, a long time. This will obviously depend on the specific test process that you participate in. In some cases, you’ll just need to spend one (or even zero) nights in the hospital. My max was 8 days and nights in a row in the hospital (including during my birthday). Not so easy for someone who prefers more freedom and their own bed…
- You’ll get used to needles, IVs, injections, and even worse things. To properly study your body’s reaction, a good amount of blood will have to be taken from you. They can do this with a small needle or take blood from you constantly through an IV (a plastic bag that will stay attached to your arm poking into one of your veins) for almost all your tests. For one of the tests I did, I stayed with a tube going through my nose to my stomach for 8 days in a row. Not that pleasant. But it’s up to you if you want to do tests like this or not.
- Side effects. Personally, throughout all the tests I did, I never suffered any worrisome side effects. Everything went smoothly and nobody had any complaints. But, of course, they do happen. Unfortunately, I wasn’t able to find the percentage of people who participate in medical trials that have undesirable side effects.

Always hooked up during the trials
Is it safe to be a guinea pig in medical trials?
This is a question that’s difficult to answer with any kind of certainty, whether positive or negative. That’s because it will depend a lot on the credentials of the lab that’s doing the study, what the drug/medicine is, and what phase the testing is in. Obviously, if you are testing a drug in phases 2,3, or 4, that means other people have already tested them. And if they are still continuing the tests, this means that the sides effects haven’t been too bad so far.
Personally, I was a little suspicious if a medicine that I was going to test had really NEVER been tested on anyone before and I would literally be the FIRST one to test it. Of the 3 times I tested drugs, luckily another group had already been tested, even if it was just the first batch of subjects from phase 1.
All of this is because certain treatments can have unpleasant or even serious side effects. Much of the time they are temporary and finish when the treatment does. Others, however, can be permanent. Some side effects appear during treatment and others don’t appear until the study has finished.
The risks also depend on the health of the person participating in the study. All of the known risks should be completely explained by researchers before the tests begin. If new information about the risks because available during the experiment, the participants should be advised.
As a positive example of someone who tested medicines, we have Robert Rodriguez, an American director that directed the films Sin City and From Dusk Till Dawn. In the beginning of his career, he tested drugs to be able to save money to finance his first films…which clearly worked well for him.
On the other side is the famous 2006 story of the 6 healthy young Brits who tested a drug and suffered a serious reaction where their heads swelled up, ending up known as the “elephant men.”
In other words, from time to time problems do happen that are serious enough to be in the news. But if you search around, you’ll see they’re not that common.
However, if worst comes to worst, the research company will usually be responsible for covering any future medical costs that you need related to the study.

Some of the other “guinea pigs” during one of my trials
How much money can I make working as a guinea pig testing drugs?
This’ll depend on the type of study. Some studies pay hundreds or even thousands of dollars to their participants.
But don’t forget the you’ll have to be able to have various weekly visits, a bunch of blood work, take medicines, and deal with possible side effects. And some of these studies can last months or even years.
And be careful, the more money you are offered, the more dangerous (or complicated or long) a study could be. But if you have any sort of illness, this could be a good opportunity to earn money, help others, and help yourself.
There are hundreds of tests for almost all types of illness and issues you can imagine. There are medical trials for people who want to stop smoking or using illicit drugs. There are also medical trials for people with allergies, mental disorders, or even for people want to lose weight. If you are interested in participating in a medical trial, do you research before applying for a study.
Where and how can I work as a guinea pig testing drugs?
If you are looking for how to do this in Brazil, please check our Portuguese page here because we have a specific list of medical trials only for Brazilians.
No matter what company you do tests for, the first step will be the same – sign up in the database of a pharmaceutical company, research center, or university. Checking out notice boards around college campuses is also an option (I’ll explain more about this below).
As I mentioned before, I tested medicines in Australia so that’s the market I’m most familiar with. However, you can find similar opportunities in most countries. If you can work legally in the country you are in, you can probably volunteer as a guinea pig to get some cash. Here are some links to browse for places to sign up around the world:
Australia
- GSK – GlaxoSmithKline (the one I worked with)
https://www.gsk-clinicalstudyregister.com
Germany
https://www.parexel.com/company/volunteer/locations-worldwide/berlin/en/trial-listings
South Africa
https://parexel.com/trials/locations/bloemfontein/en
United States/Canada
https://www.parexel.com/company/volunteer
https://www.covanceclinicaltrials.com/en-us/
- National Institute on Drug Abuse
https://www.drugabuse.gov/clinical-trial/search
- National Cancer Institute
https://www.cancer.gov/about-cancer/treatment/clinical-trials
- National Institute of Mental Health
https://www.nimh.nih.gov/health/trials/index.shtml
- National Institute of Health
https://www.nih.gov/health-information/nih-clinical-research-trials-you/finding-clinical-trial
https://clinicalstudies.info.nih.gov

Playing video games while in the hospital during my medical trial
- Donating Plasma
Getting paid to donate plasma in the United States is actually something very common. I met a few people doing it and they said it was easy.
Basically, it’s like giving blood but after the blood leaves your body, it goes through a machine that takes out the plasma and “returns” the blood back to you.
One session is about 90 minutes and you can donate up to 2 times a week. The price for donating depends on the place, but I’ve seen payments of USD $50 for a donation.
There’s a lot of plasma donation centers spread throughout the United States. This website below can help you find one close to you.
United Kingdom (England, Scotland, Wales, and Northern Ireland)
- GSK – GlaxoSmithKline
https://volunteers.gsk.co.uk/our-trials.aspx
- Others in the UK
https://www.drugtrial.co.uk/en/volunteer-info/
https://www.nhs.uk/Conditions/Psoriasis/Pages/clinical-trial.aspx
Around the world!
I think these three websites below are the best to find medical trials wherever you are in the world:
- CenterWatch.com
https://www.centerwatch.com/clinical-trials/listings/locations.aspx
This website is one of the biggest and most thorough databases online with global listings for clinical studies sponsored by the industry and governments worldwide. They also have a blog and an entire section on the site that tells you everything you need to know about volunteering for a medical trial.
- ClinicalTrials.gov
Another really good database with clinical studies worldwide.
- CISCRP – The Center for Information and Study on Clinical Research Participation
https://www.centerwatch.com/directories/1068-useful-resources/listing/2826-clinical-research-community-by-ciscrp
Yet another gigantic database to volunteer for medical trials.
University notice boards around the world
This is a tip to use for anywhere in the world. I’ve visited a lot of universities in other countries where the notice boards have postings in search of volunteers to participate in medical trials or other (less invasive) research studies.
At Harvard (no, I wasn’t a student…just visiting :-), I saw a bunch of notices looking for people to spend several days sleeping in special rooms to evaluate something related to the exposure of a certain light.
Personally, I participated in two studies I found at universities. At the University of Minnesota, I spent a few hours inside an MRI machine receiving small shocks on my big toe while seeing figures on a screen. Not a big deal and I received about $50 for it.
At James Madison University, I had to wear a special watch to evaluate my sleep over the course of a week. It was super easy.
So no matter where you are in the world, don’t forget to visit some of the large universities nearby to see if there are any opportunities to make a little bit of extra cash.

This is the kind of university notice board I’m talking about. Picture by Ken Banks, licensed under CC BY 2.0

Picture by Bala Sivakumar, licensed under CC BY-SA 2.0

Example #1 of study I found at Harvard

Example #2 of study I found at Harvard – it’s been several years, so I recommend you don’t try calling that number
Ready to get started testing medicines?
Testing medicines really isn’t for everyone, whether in health or psychological terms. It might also be hard to deal with the judgment of others if you are a healthy person testing medicines (though not as much if you are actually sick and trying to find a cure).
In the first case, the hypocrisy is obvious because if there weren’t volunteers willing to try medicines, research for new drugs would stop. Then, there wouldn’t be new medicines to try to improve the population’s general health and increase the quality of life.
On that note, if you’ve already decided that you want to test medicines, here are some tips:
- Learn about and understand the different types of medical research and studies
- Know what questions to ask and what your rights are as a participant
- Discover what the possible risks are and decide what level of risk you are willing to take before signing up for medical trials.
Okay, well that was a long article but I hope it touched on all the important points. But in case you have any more questions that I can help with, just let me know in the comments area below!
Planning your next trip?

Already reserved your hotel or hostel? If not, our article with The 6 Best and Cheapest Websites to Find & Reserve Accommodation can help you out. You’ll also find some promotions and discount codes.
Still haven’t booked your plane ticket and want to save big? Take a look at our page with 16 Tips to Save on Flights where you’ll also find the 4 best websites to buy your plane tickets.
And finally, will you need to rent a car during your trip? Then surely our page with The 5 Best and Cheapest Websites to Compare and Rent Cars Around the World will help you choose the best rental car and find a good deal.




 8LvG4w56H8xI6oS3MDSK2mZ1enEH_ABlpTx5jAsYdFQ
8LvG4w56H8xI6oS3MDSK2mZ1enEH_ABlpTx5jAsYdFQ